DEHP
A Green PVC? Not So Fast, Many Problems Still Remain
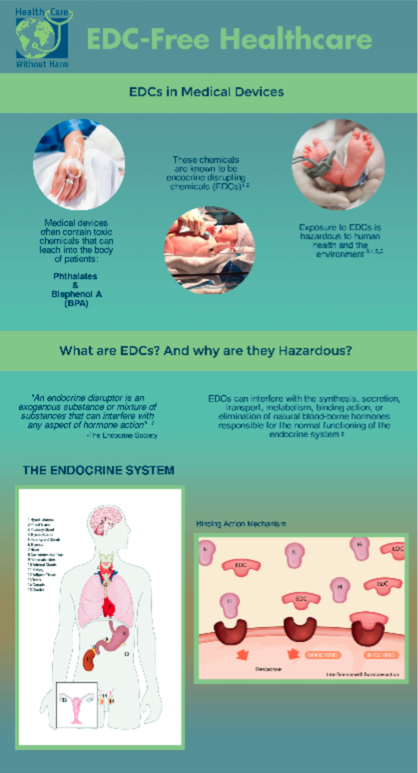
EDC-Free Healthcare Infographic
Joint NGO Letter to MEP Glenis Willmott Regarding Medical Device Regulation Amendment
On November 11th 2015, HCWH Europe sent joint letter, co-signed by 18 different environmental and health NGOs, to MEP Glenis Willmott, seeking her support for an amendment to the European Commission (EC) Regulation Proposal on Medical Devices in trialogue negotiations between the European Parliament, the Luxembourg and Dutch Presidencies of the European Council, and the European Commission. Read the open letter here.
18 NGOs Co-Sign Letter to MEP regarding Upcoming Medical Device Regulation Negotiations
DEHP: A Threat to Male Fertility
Prenatal exposure to phthalate DEHP affects development of baby boys
DEHP Authorisation - Joint Letter to European Commission: Briefing Document (January 2015)
Briefing document in connection with the Joint Letter to European Commission on the authorisation of DEHP in PVC plastic. This outlines procedural and substantive flaws of the DEHP in PVC opinions.
Released: January 2015
DEHP Authorisation - Joint Letter to European Commission (January 2015)
Joint letter from European civil society organisations asking the European Commission to reject authorisation of the substance of very high concern DEHP in PVC plastic. Briefing document also available.
Signed by Health Care Without Harm (HCWH) Europe in January 2015.
Press release | Premature infants are exposed to unsafe levels of chemical in medical products used to save their lives

Johns Hopkins Bloomberg School of Public Health
Páginas
- « primera
- ‹ anterior
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- siguiente ›
- última »